Oral Microbiota-Based Therapeutics: TranslatingFunctional Microbiome Science into Patient-Centric Delivery
Introduction
Microbiome therapeutics have progressed from experimental interventions to regulated medicinal products. Functional and spatial microbiome research has clarified how microbial communities influence host immunity, metabolism, and drug response, while emerging Good Manufacturing Practice (GMP) frameworks enable standardised production of live biotherapeutic products (LBPs).
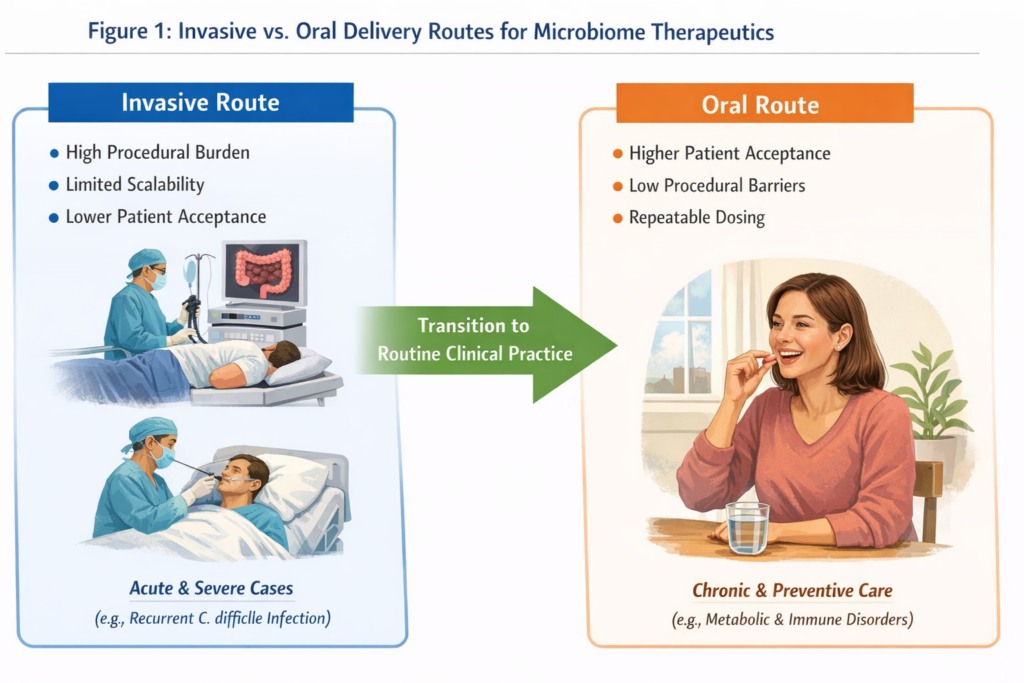
Despite this progress, a central translational question remains: how do microbiome therapeutics reach patients in a scalable and acceptable way? Early clinical success relied largely on invasive fecal microbiota transplantation (FMT), effective in select indications but limited by procedural burden, scalability, and patient acceptance [1, 2]. Delivery strategy has therefore emerged as a key bottleneck for broader clinical adoption.
From Functional Insight to Delivery Constraints
Microbiome function is shaped by spatial localization, host-microbe interfaces, and intestinal microenvironments. Therapeutic efficacy depends not only on microbial composition, but on whether functional activity is delivered to relevant niches in vivo. As a result, delivery becomes a biology-aware challenge rather than a purely pharmaceutical one.
Oral microbiota-based therapeutics must survive gastric transit, reach target intestinal regions, and maintain functional activity long enough to exert benefit. Approaches that ignore these constraints risk reduced efficacy, even when manufacturing quality is high.
GMP as an Enabler of Oral Delivery
GMP standardization plays a central role in enabling patient-friendly delivery. Controlled donor screening, reproducible manufacturing, validated potency metrics, and stability testing collectively reduce safety risks and batch variability. These advances support formulation strategies such as encapsulation and lyophilization, making oral administration feasible beyond specialized hospital procedures.
In this context, GMP does not merely support regulatory compliance; it expands the design space for scalable and acceptable delivery.
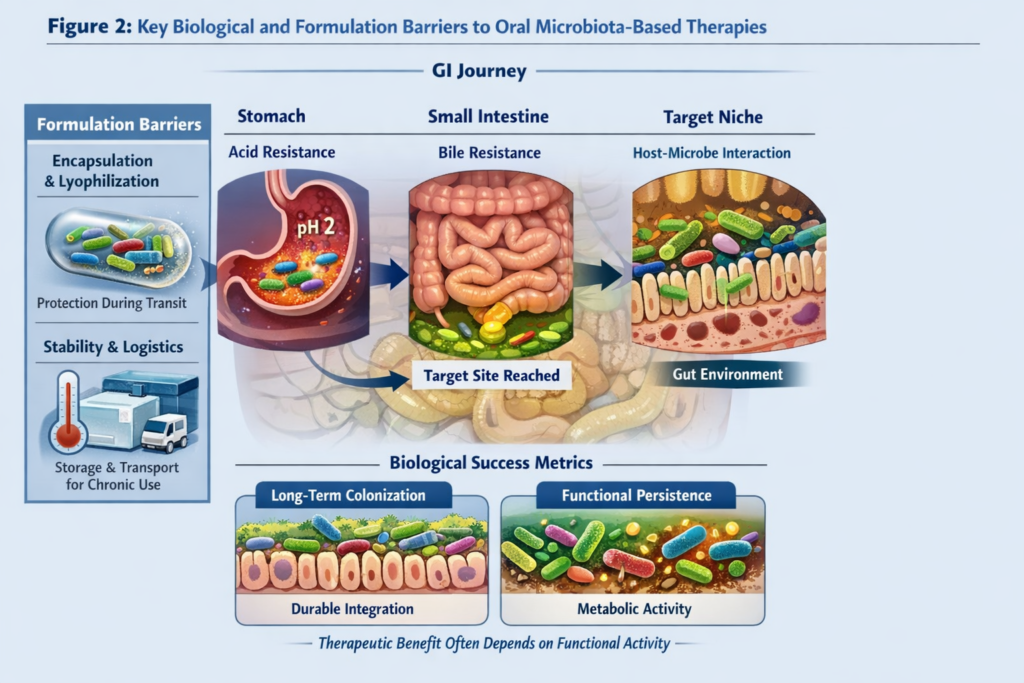
Scientific Challenges of Oral Microbiome Therapeutics
Oral delivery of live microbial therapies presents several scientific challenges:
- Survival through the gastrointestinal tract, including resistance to acid and bile.
- Functional persistence, as therapeutic benefit may depend on metabolic activity rather than long-term colonisation [3].
- Dose-response relationships, complicated by inter-individual variability in microbiome composition and host physiology [4].
- Stability and logistics, critical for outpatient and chronic use.
Addressing these challenges requires integrating microbiology, formulation science, and clinical insight, with a focus on preserving function rather than on taxonomy alone.
Clinical Expansion Beyond Acute Indications
FMT-based approaches demonstrated strong efficacy in acute and recurrent Clostridioides difficile infection [5]. However, extending microbiome therapeutics to chronic inflammatory, metabolic, and immune-mediated diseases requires delivery models compatible with repeated dosing and outpatient care.
Oral formulations lower procedural barriers and improve patient acceptance, enabling longer treatment durations and broader populations. This shift is essential for translating microbiome science into indications where sustained modulation, rather than acute intervention, is required. While invasive delivery routes remain clinically necessary in acute and severe settings, oral formulations are essential for expanding microbiome therapeutics to chronic, outpatient, and preventive indications.
Role of Clinical Networks
Hospitals and clinical networks remain essential for the deployment of microbiome therapeutics. Beyond clinical trials, they enable patient stratification, longitudinal monitoring, and real-world evidence generation. Feedback from clinical practice informs formulation refinement, dosing strategies, and regulatory dialog, reinforcing delivery as an iterative component of translation rather than a final step.
Outlook
The evolution of microbiome therapeutics reflects a broader shift toward function-driven and patient-centric medicine. Functional and spatial insights define what biological signals matter, GMP enables reproducible manufacturing, and delivery determines whether therapies can integrate into real-world care.
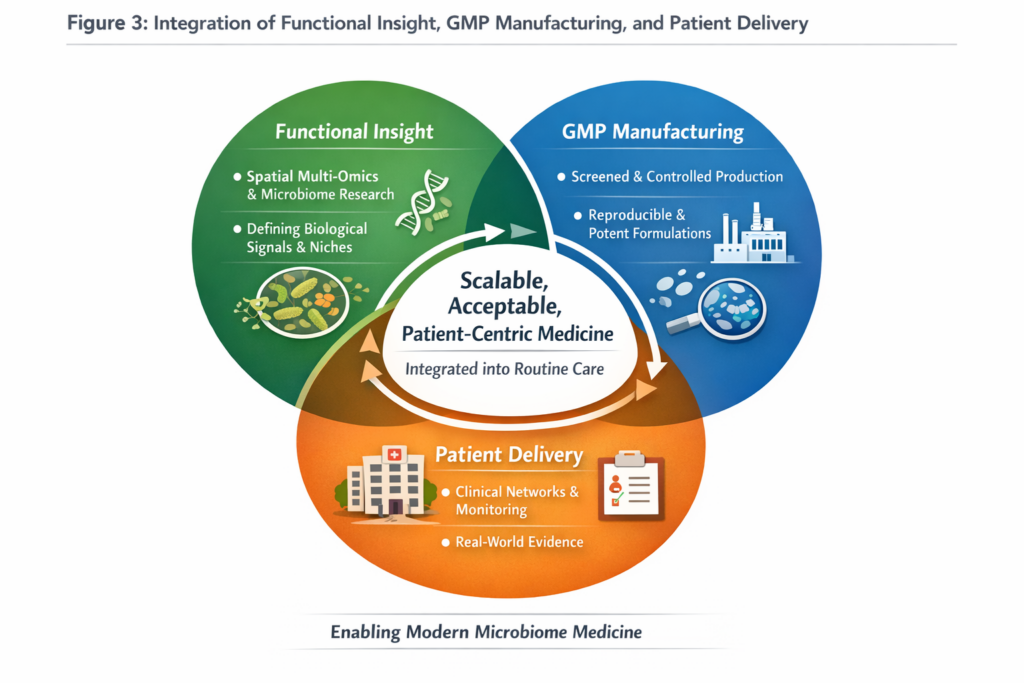
Scalable and acceptable delivery strategies will ultimately determine whether microbiome therapeutics transition from specialized interventions to routine clinical practice. Patient-centric delivery is therefore not ancillary to microbiome innovation; it is a prerequisite for its success.
Together with spatial multi-omics insights and standardized GMP frameworks discussed previously, scalable delivery strategies are the final translational piece enabling microbiome therapeutics to become part of routine patient care.
References
[1] Alexander Khoruts and Michael J. Sadowsky. “Understanding the mechanisms of faecal microbiota transplantation”. In: Nature Reviews Gastroenterology & Hepatology 13 (2016), pp. 508–516. doi:10.1038/nrgastro.2016.98.
[2] Jonathan Chen et al. “Stool Banking for Fecal Microbiota Transplantation”. In: Frontiers in Cellular and Infection Microbiology 11 (2021). doi: 10.3389/fcimb.2021.622949.
[3] John F. Cryan et al. “The Microbiota–Gut–Brain Axis”. In: Physiological Reviews 99.4 (2019), pp. 1877–2013. doi:10.1152/physrev.00018.2018.
[4] Niv Zmora, Jotham Suez, and Eran Elinav. “You are what you eat: diet, health and the gut microbiota”. In: Nature Reviews Gastroenterology & Hepatology 16 (2019), pp. 35–56. doi:10.1038/s41575-018-0061-2.
[5] Stefano Di Bella, Theodore Gouliouris, Nicola Petrosillo, et al. “Fecal microbiota transplantation (FMT) for Clostridium difficile infection: Focus on immunocompromised patients”. In: Journal of Infection and Chemotherapy 21.4 (2015), pp. 230–237. doi:10.1016/j.jiac.2015.01.011.
